
Note: This video is a good one to watch while reading the Introduction of the Science of Ionized Water.
A comprehensive scientific exploration of ionized water, molecular hydrogen, water structure, electrolysis, and emerging research into the physical and biological properties of water.
Introduction
Water is the most familiar substance in our daily lives.
It fills our oceans, flows through rivers, falls from the sky, and makes up a large portion of the human body. Yet despite centuries of scientific investigation, water remains one of the most complex and fascinating substances on Earth.
Researchers studying water have discovered that even small changes in its chemistry — including dissolved gases, electrical charge, mineral balance, and electrochemical conditions — may influence how water interacts with the body and its potential health effects.
In recent decades, two technologies have brought these questions into everyday conversation:
- water ionizers
- hydrogen water machines
Both alter water in measurable ways and have attracted growing scientific interest about their benefits.
Some discussions focus heavily on alkaline pH. Others emphasize molecular hydrogen. Still others explore more experimental ideas about water structure and energetic properties. Without a basic understanding of the science, the subject can quickly become confusing.
This page was created to explore these topics in a clear, thoughtful way and to give general readers a stronger foundation for understanding the science behind them.
Rather than reducing the subject to simple claims or dismissing emerging ideas too quickly, the goal here is to examine the science step by step.
Along the way we will explore:
- how water ionizers split water into two streams
- what molecular hydrogen research has discovered
- why even small doses of hydrogen may influence cellular signaling
- how electrochemistry changes water chemistry
- what scientists are investigating about the structure of water and its impact on health
Some of these topics are well established.
Others remain areas of active research.
Understanding the difference between the two types of water machinies is part of understanding the science itself.
For over three decades we have studied water ionizers, filtration systems, and hydrogen water technologies. This page brings together both scientific research and long-term practical experience working with these systems.
How to Use This Page
This page is designed as a guided exploration rather than a short article.
Each section builds on the previous one, gradually expanding the reader’s understanding of how water chemistry, electrolysis, and hydrogen research intersect.
If you prefer to jump directly to a topic of interest, use the navigation links below. Otherwise, reading the sections in order will provide the clearest overview of the subject.
Jump to Section
- Section 1: Why Water Science Matters & Molecular Hydrogen and Cellular Signaling
- Section 2: How Water Ionizers Split Water into Two Streams
- Section 3: Small Doses, Regular Intake, and Why Consistency Matters
- Section 4: Protective Pathways Inside the Body
- Section 5: Water Structure and Where the Science is Still Evolving
- Section 6: Electrochemistry and Platinum Catalysis
- Section 7: Ionized Water vs. Hydrogen Water Machines
- Section 8: Measuring Hydrogen in Water
Section 1: Why Water Science Matters & Molecular Hydrogen

Note: This video will help to understand and visualize this section of the Science of Ionized Water
Water is the most abundant molecule in the human body and the foundation of nearly every biological process.
Every cell depends on water to transport nutrients, regulate temperature, remove metabolic waste, maintain electrical balance, and support cellular chemistry.
Yet water is often treated as though all forms are essentially the same.
For many years, water research focused mainly on purity and contamination. Questions about bacteria, heavy metals, pesticides, and industrial chemicals rightly became major public health concerns.
But over the past several decades, researchers have also begun asking another question:
Could the physical and chemical characteristics of water itself influence biological systems?
That question has helped drive growing interest in several areas of research, including:
- molecular hydrogen in water
- electrochemistry and ionized water
- dissolved gas dynamics
- mineral balance and buffering capacity
- possible structural organization of water molecules
Two technologies that intersect with this research are water ionizers and hydrogen water machines.
Both technologies can alter water and its benefits in measurable ways.
The Discovery That Changed Hydrogen Science
For much of the twentieth century, molecular hydrogen was considered biologically inert.
It was assumed to pass through the body without doing anything significant.
That assumption changed in 2007, when a research team led by Dr. Shigeo Ohta published a landmark study showing that hydrogen gas could reduce oxidative damage in cells.
Study:
Ohta et al.
Hydrogen acts as a therapeutic antioxidant
Nature Medicine (2007)
https://www.nature.com/articles/nm1577
This discovery helped launch what is now often called hydrogen medicine.
Since then, hundreds of studies have explored the possible biological effects of molecular hydrogen.
What Is Molecular Hydrogen?
Molecular hydrogen is the simplest molecule in the universe.
It consists of two hydrogen atoms bonded together: H2.
Because hydrogen is extremely small and electrically neutral, it can move rapidly through biological tissues.
Researchers became especially interested in hydrogen because it can diffuse through:
- cell membranes
- mitochondria
- the blood-brain barrier
This ability to move quickly through the body and boost its protective pathways helps explain why hydrogen drew scientific attention in the first place.
Why This Matters
The discovery that dissolved hydrogen may benefit biology opened a new line of inquiry in water science.
It suggested that water could be more than a delivery vehicle for nutrition or waste removal. Under some conditions, its dissolved gases and electrochemical properties might also matter.
This is one reason water ionizers and hydrogen water machines have received growing interest. Both are based, in part, on the idea that changing water chemistry may affect more than purity or taste alone.
The next question, of course, is how a tiny molecule like hydrogen is produced in water in the first place. That begins with understanding how a water ionizer works which we'll explore in the next section.
Further Reading
- Ohta et al. (2007)
Hydrogen acts as a therapeutic antioxidant
Nature Medicine
https://www.nature.com/articles/nm1577 - LeBaron et al. (2022)
Electrolyzed Reduced Water and Molecular Hydrogen
International Journal of Molecular Sciences
https://www.mdpi.com/1422-0067/23/19/11257 - Ichihara et al. (2015)
Beneficial biological effects of molecular hydrogen
Medical Gas Research
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4488660/
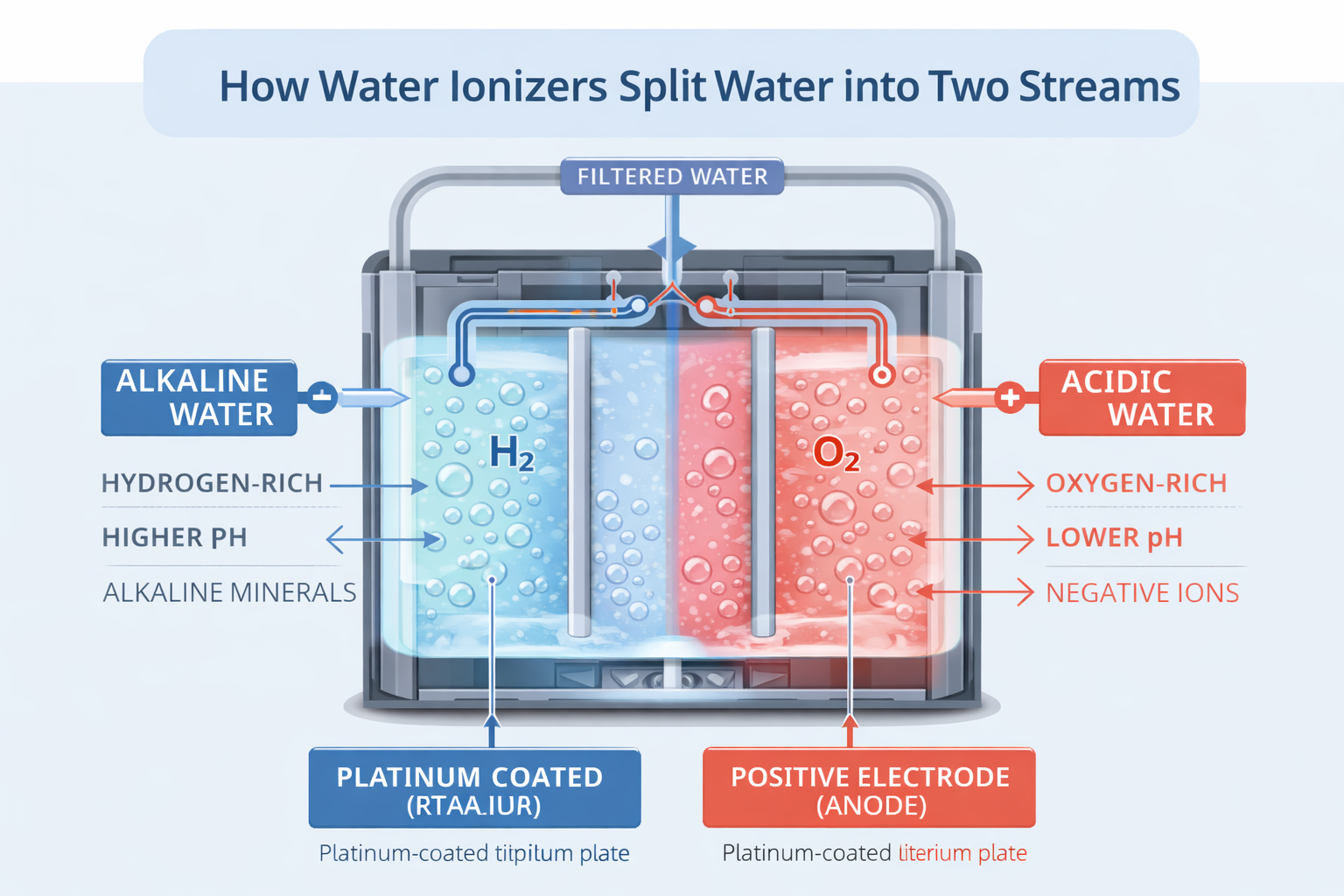
Section 2: How Water Ionizers Split Water into Two Streams
Note: This video helps visualizing the workings of a water ionizer and getting a little more data about it.
One of the most distinctive things about a water ionizer is that it does not simply clean water.
It changes water through a process called electrolysis, creating two separate streams with different characteristics.
A standard filter removes unwanted substances. A water ionizer begins with filtration, but then goes further by passing water across electrically charged plates that alter its chemistry as it flows through the machine.
At the simplest level, the process can be summarized like this: A water ionizer uses electricity to separate water into an alkaline stream and an acidic stream.
What Happens Inside a Water Ionizer?
Inside a typical water ionizer, incoming tap water first passes through a filter.
This matters because electrolysis works best on water that has been cleaned of common contaminants while still retaining enough dissolved minerals to conduct electricity.
After filtration, the water enters an electrolysis chamber containing metal plates. Even in average quality water ionizers, these plates are made of titanium coated with platinum.
When electrical current passes through those plates, the water and dissolved minerals begin to respond.
Several things happen at once:
- Water participates in electrochemical reactions, and dissolved mineral ions separate into two streams.
- Hydrogen gas is generated at the cathode side (the negatively charged electrode).
- Hydroxide ions become more concentrated in the alkaline stream.
- Alkaline minerals such as calcium, magnesium, and potassium become more concentrated on the alkaline side, while acid-forming ions and more oxidative conditions increase on the acidic side. The acidic water also has practical uses. See the Water Ionizer Use and Care page.
The Two Streams: Alkaline and Acidic
The result is two distinct output streams coming out of the water ionizer.
One is the alkaline stream, which is most commonly used for drinking and cooking. It typically contains a higher pH and dissolved molecular hydrogen.
The other is the acidic stream, which typically has a lower pH, greater oxidizing potential (for sanitizing), and different household uses than the drinking stream.
Many water ionizers have separate settings for drinking water and acidic water output, but these are not usually necessary as the two streams come out simultaneously. Machines usually have several settings, ranging from light to strong ionization, to accomodate the needs and wants of customers.
The Basic Chemistry Behind the Alkaline Side
At the cathode side of the ionizer, one of the main reactions is commonly represented as:
2H2O + 2e- → H2 + 2OH-
This equation helps explain two important results of electrolysis.
First, it shows why molecular hydrogen is produced.
Second, it shows why the water becomes more alkaline, because hydroxide ions increase on that side of the chamber.
This is one reason older discussions of ionized water often focused mainly on pH and ORP (oxidation reduction potential), while newer discussions place more emphasis on dissolved hydrogen.
Why Minerals Matter
Electrolysis depends on conductivity, and conductivity depends largely on dissolved minerals in the water.
If water is too low in minerals, electrolysis becomes less efficient. This is why some ionizers do not perform well with very low-mineral water unless mineral content is adjusted.
Then on the oposite end, water can contain too many highly conductive minerals when it is using water that has been through a sodium exchange softener. These issues are discussed and explained on other pages, such as the Water Ionizer Use and Care Page.
Common dissolved minerals that contribute to conductivity include calcium, magnesium, potassium, and sodium. These minerals influence the pH range the machine can produce.
When water leaves the electrolysis chamber, it is measurably different from the filtered tap water that entered the machine.
Depending on the machine, source water, and settings, the alkaline drinking water may differ in taste or feel, in addition to potential health benefits, compared with ordinary filtered water.
Plain English Takeaway
A water ionizer filters water and then uses electricity to separate it into two different streams.
One becomes the alkaline drinking water most people associate with ionizers. The other becomes an acidic stream with its own uses.
During this process, the water changes in several measurable ways, including pH, hydrogen production, mineral balance, and electrochemical behavior (with a negative charge and greater concentration of calcium, magnesium and other healthy minerals in the alkaline stream).
This helps explain why ionized water is not simply filtered water under another name.
Further Reading
- LeBaron et al. (2022)
Electrolyzed Reduced Water: Review I. Molecular Hydrogen Is the Exclusive Agent Responsible for the Therapeutic Effects
International Journal of Molecular Sciences
https://www.mdpi.com/1422-0067/23/19/11257 - Shirahata et al. (2012)
Electrolyzed-Reduced Water Scavenges Active Oxygen Species and Protects DNA from Oxidative Damage
Biochemical and Biophysical Research Communications
https://pubmed.ncbi.nlm.nih.gov/15163546/
Section 3: Small Doses, Regular Intake, and Why Consistency Matters

Note: I suggest watching this video along with reading this section. It helps in clarification.
One of the most surprising findings in molecular hydrogen research is that very small amounts of hydrogen may produce measurable biological responses.
This can seem counterintuitive at first.
In chemistry, we often assume that stronger effects require larger quantities. But biological signaling systems do not always work that way. In living systems, small triggers can activate much larger protective responses.
Hormones, neurotransmitters, and other signaling molecules often operate in extremely small amounts. Hydrogen appears to be important for similar reasons.
Rather than functioning only as a bulk chemical antioxidant, hydrogen may act more like a signal that helps the body activate its own defense systems.
Why Hydrogen Does Not Need to Stay in the Body for Long
Hydrogen molecules are extremely small and diffuse rapidly through tissues.
After hydrogen-rich water is consumed, hydrogen levels typically rise quickly and then gradually decline as the gas is exhaled through the lungs.
In many cases, hydrogen levels return toward baseline within about an hour or two.
But if hydrogen acts as a signaling molecule, its brief presence may still be enough to trigger beneficial cellular responses that continue afterward.
A simple analogy is a light switch. The switch is touched only briefly, but once the circuit is activated, the light remains on.
The Importance of Consistency
Because hydrogen does not remain in the body very long, many researchers believe regular exposure may matter more than occasional large doses.
Instead of emphasizing extremely high hydrogen concentrations all at once, many studies focus on the effects of drinking hydrogen-rich water consistently over time.
This fits naturally with normal hydration habits. Most people do not drink all of their daily water at once. They drink it gradually throughout the day.
Water Ionizers and Fresh Hydrogen Production
Water ionizers are especially well suited to this pattern because they produce hydrogen-rich water on demand.
Each time fresh water is drawn from the machine, electrolysis produces dissolved hydrogen in the water.
This allows hydrogen-rich water to be consumed throughout the day as part of a regular hydration routine.
Why Extremely High Concentrations May Not Be Necessary
If hydrogen primarily works through signaling pathways, then modest concentrations consumed regularly may be enough to stimulate those pathways.
This helps explain why many long-term hydrogen studies use moderate hydrogen levels combined with consistent daily intake rather than extremely high concentrations taken occasionally.
Plain English Takeaway
Hydrogen does not need to remain in the body for hours to matter.
Its value may lie in briefly triggering protective responses inside cells.
That is why regular intake of freshly produced hydrogen-rich water may be more important than chasing extreme concentrations.
Small amounts, consumed consistently, may be enough.
Section 4: Protective Pathways Inside the Body

Note: Watch this video for a greater unserstanding of this section.
How Cells Respond to Stress, Signals, and Repair
Living cells are constantly responding to their environment.
Every second, trillions of chemical reactions occur inside the body. These reactions generate energy, build molecules, repair tissues, and maintain the balance required for life.
At the same time, these reactions also produce reactive oxygen species (ROS), often called free radicals.
In small amounts, ROS are a normal part of cellular communication. Problems arise when too many accumulate and the body's protective systems cannot keep up.
This imbalance is commonly referred to as oxidative stress.
The Body's Built-In Defense System
Cells are not defenseless against oxidative stress. They contain internal systems designed to restore balance.
One of the most important systems studied in hydrogen research is the Nrf2 pathway.
Nrf2 stands for Nuclear factor erythroid 2–related factor 2. The name is technical, but the basic idea is simple: Nrf2 acts like a master switch that tells cells to turn on their internal defense and repair systems.
When cells detect stress, Nrf2 moves into the nucleus and signals specific genes to begin producing protective enzymes.
These enzymes help the body:
- neutralize excess free radicals
- reduce oxidative stress
- repair damaged molecules
- maintain cellular balance
A Helpful Analogy
One way to picture the Nrf2 pathway is to think of it as a fire alarm system inside the cell.
When cellular stress rises, the alarm is triggered. Once that happens, protective responses are deployed.
The enzymes associated with this pathway include several of the body's most important antioxidant defenses:
- superoxide dismutase
- catalase
- glutathione-related enzymes
These are part of the body's natural defense network, working continuously to help keep oxidative stress under control.
Hydrogen and the Activation of Protective Pathways
This is where molecular hydrogen becomes especially interesting.
Research suggests that hydrogen may influence signaling pathways that help regulate systems like Nrf2.
Rather than acting only as a direct antioxidant, hydrogen appears to function more like a regulatory signal that encourages cells to activate their own protective systems.
Studies suggest hydrogen exposure may:
- stimulate antioxidant enzyme production
- reduce inflammatory signaling
- support mitochondrial balance
- help regulate cellular stress responses
Because these responses involve gene activation and enzyme production, their effects can continue after the hydrogen molecules themselves have dissipated.
The Role of Mitochondria
Mitochondria are often described as the power plants of the cell because they generate the energy needed for almost every biological process.
During energy production, they naturally generate reactive oxygen species as byproducts of metabolism.
Under normal conditions this is not harmful. Cells are designed to manage these molecules.
Hydrogen research suggests molecular hydrogen may help support the balance between energy production and oxidative stress within mitochondria.
Why This Matters
Health depends on balance.
The body constantly regulates opposing forces such as:
- oxidation and antioxidant defense
- inflammation and resolution
- energy production and repair
The goal is not to eliminate reactive molecules entirely. The goal is to maintain equilibrium.
This helps explain why researchers view molecular hydrogen as more than a simple antioxidant. Its possible importance may lie in how it influences the body’s own protective response systems.
Section 5: Water Chemistry, Emerging Questions, and Where the Science Is Still Evolving

Why Water Still Deserves Serious Study
Water may seem simple, but in reality it is one of the most unusual and important substances in biology.
Every molecule of water is made of just two hydrogen atoms and one oxygen atom. Yet when countless water molecules interact with one another, the result is a liquid with remarkable properties that support life itself.
Water helps regulate temperature, dissolve nutrients, transport minerals, remove waste, support electrical signaling, and serve as the medium in which nearly all cellular chemistry takes place.
Because of this, scientists do not study water only as something we drink. They study it as the chemical environment in which life happens.
Why Water Behaves Differently Than Many Other Liquids
Water molecules are constantly interacting through hydrogen bonds. These are temporary attractions between molecules that form and break rapidly, creating a dynamic network rather than a rigid structure.
This helps explain why water has several unusual properties, including:
- high surface tension
- high heat capacity
- strong dissolving ability
- an exceptional ability to support biological reactions
In plain English, water is not just a neutral backdrop. It is an active part of the chemical environment of the body.
Why Modern Water Research Has Expanded
For many years, most public discussions about water focused mainly on purity. That made sense, because removing contaminants remains one of the most important goals in drinking water.
But more recent research has expanded the conversation.
Scientists now also study how changes in water chemistry may matter, including changes in:
- dissolved gases
- mineral balance
- buffering behavior
- electrical and electrochemical conditions
- oxidation-reduction characteristics
This is where technologies such as water ionizers and hydrogen water machines become especially relevant. These systems do more than filter water. They can also change measurable chemical properties of the water.
Dissolved Gases Matter More Than Many People Realize
One of the biggest developments in recent years has been the growing recognition that dissolved gases (like H2) can influence biological systems.
Molecular hydrogen is the clearest example discussed on this page, but it is part of a broader scientific principle: water can carry dissolved gases that affect how the water behaves and how it interacts with the body.
This is one reason hydrogen water research has gained so much attention. It highlights the fact that water is not defined only by H2O itself, but also by what is dissolved in it and how that water has been processed.
Mineral Balance and Buffering Capacity
Another area of growing interest is the role of mineral composition.
Dissolved minerals such as calcium, magnesium, potassium, and sodium influence much more than taste. They also affect conductivity, buffering capacity, and how water behaves during electrolysis.
Buffering capacity refers to water's ability to resist sudden pH changes. This matters because water with different mineral profiles can behave differently in the body, in cooking, and in the performance of an ionizer.
So when people compare one water source to another, or compare filtered water to ionized water, they are often comparing more than purity alone. They are also comparing mineral behavior and chemical balance.
Nanobubbles and Microenvironments
Another area of ongoing research involves nanobubbles and electrochemical microenvironments.
During electrolysis, hydrogen gas forms at the electrode surfaces. Some of that hydrogen dissolves into the water, while some may initially exist as very small bubbles.
Researchers continue studying how these tiny bubbles behave, how long they persist, and whether they influence hydrogen delivery or water properties in meaningful ways.
Scientists are also interested in the highly localized chemical conditions that exist right near electrode surfaces during electrolysis. These tiny zones can differ from the bulk water and may help explain why electrochemically treated water can behave differently from water that has simply had gas infused into it.
This area of research is still developing, but it is directly relevant to understanding what happens inside a quality water ionizer.
Electrolysis Creates a Unique Chemical Environment
Water ionizers are especially interesting because they create a temporary electrochemical environment inside the machine.
During electrolysis, water is exposed to:
- electrical current
- charged electrode surfaces
- mineral migration
- dissolved hydrogen formation
- charge separation into two streams
These conditions clearly alter the chemistry of the water in measurable ways.
Some of these changes are well established, including the production of dissolved hydrogen, changes in pH, changes in oxidation-reduction potential, and redistribution of minerals between the alkaline and acidic streams.
Other questions are still being explored, including how these electrochemical conditions may influence water behavior at a more subtle level.
Here's the Million-Dollar Question I Hear All the Time: What About Stomach Acid?
One of the most common questions I hear from doctors, and sometimes from their patients, is this:
What about stomach acid? Doesn’t alkaline ionized water interfere with it?
That is the million-dollar question.
Here is the practical answer:
Any water or other beverage has the potential to temporarily affect stomach acidity if too much is consumed at once, especially with meals. This is not unique to alkaline ionized water.
For example, let’s say you drink an 8-ounce glass of distilled water with a meal. That is actually quite a bit of water to consume during eating. Even though distilled water may test around neutral pH 7.0, or sometimes slightly acidic around pH 6.0, it is still far less acidic than the stomach environment that supports digestion, which is often around pH 3.5 or lower.
So what can you do?
One simple approach is to add a squeeze of lemon to the water you consume around mealtimes. Apple cider vinegar can work similarly. This can bring the pH of the water down significantly, often to around pH 4.
Beyond that, I usually suggest not drinking large amounts of any water quickly during meals. Also, instead of gulping, just take small sips as needed.
I have done a lot of experimenting with adding lemon to different types of water, and whether the starting point is 9.5 alkaline ionized water or 6.0 distilled water, a squeeze of lemon can bring both down to around pH 4.
You can see that demonstrated in this short video clip about The Million Dollar Question about Water at Mealtimes.
Why Some Questions Are Still Open
Water is difficult to study in complete detail because it is highly dynamic.
Molecules are constantly moving. Dissolved gases can escape quickly. Electrochemical conditions can change from one moment to the next. Small differences in minerals, temperature, flow rate, and electrode design can all influence the outcome.
That is one reason water science continues to evolve.
Some questions already have strong evidence behind them. For example, molecular hydrogen has become a major focus because it can be measured, studied biologically, and connected to specific signaling pathways.
Other questions remain more open. Researchers continue investigating how dissolved gases, mineral conditions, electrolysis, and micro-scale water behavior may interact.
Why an Open but Careful Approach Matters
Good science does not require pretending that every question has already been answered. It also does not require accepting every theory uncritically.
The most responsible approach is to distinguish between what is clearly established, what is strongly supported, and what is still emerging.
At this point, several things are clear:
- water chemistry matters
- dissolved molecular hydrogen is biologically relevant
- electrolysis changes water in multiple measurable ways
- mineral balance and water quality affect how these systems perform
Beyond that, science is still exploring how all these factors may fit together in the full picture of water and health.
Why This Matters in Daily Life
For the average person, all of this comes back to a practical question: does the way water is produced matter?
The growing body of research suggests that it can.
Clean water remains the first priority. But once water is clean, other characteristics — such as dissolved hydrogen, mineral balance, and electrochemical properties — may also influence how the water behaves and how people experience it in daily life.
This helps explain why some people notice meaningful differences between ordinary filtered water, hydrogen water, and freshly produced ionized water.
Conclusion
Water science is still expanding.
The more researchers study water, the clearer it becomes that water is not just a passive liquid carrying nutrients from one place to another. It is part of a dynamic chemical environment that supports every function of life.
Some parts of this science are now well established. Other parts are still unfolding. But the direction of the research is clear: water deserves far more attention than it once received.
Understanding water chemistry, dissolved hydrogen, mineral behavior, and electrolysis gives us a deeper and more useful framework for thinking about hydration, balance, and the technologies designed to improve the water we drink every day.
Further Reading
- LeBaron et al. (2022)
Electrolyzed Reduced Water: Review I. Molecular Hydrogen Is the Exclusive Agent Responsible for the Therapeutic Effects
International Journal of Molecular Sciences
https://www.mdpi.com/1422-0067/23/19/11257 - Ichihara et al. (2015)
Beneficial Biological Effects and the Underlying Mechanisms of Molecular Hydrogen
Medical Gas Research
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4488660/ - Ohta (2014)
Molecular Hydrogen as a Preventive and Therapeutic Medical Gas: Initiation, Development and Potential of Hydrogen Medicine
Pharmacology & Therapeutics
https://pubmed.ncbi.nlm.nih.gov/24316428/
Section 6: Electrochemistry and Platinum Catalysis
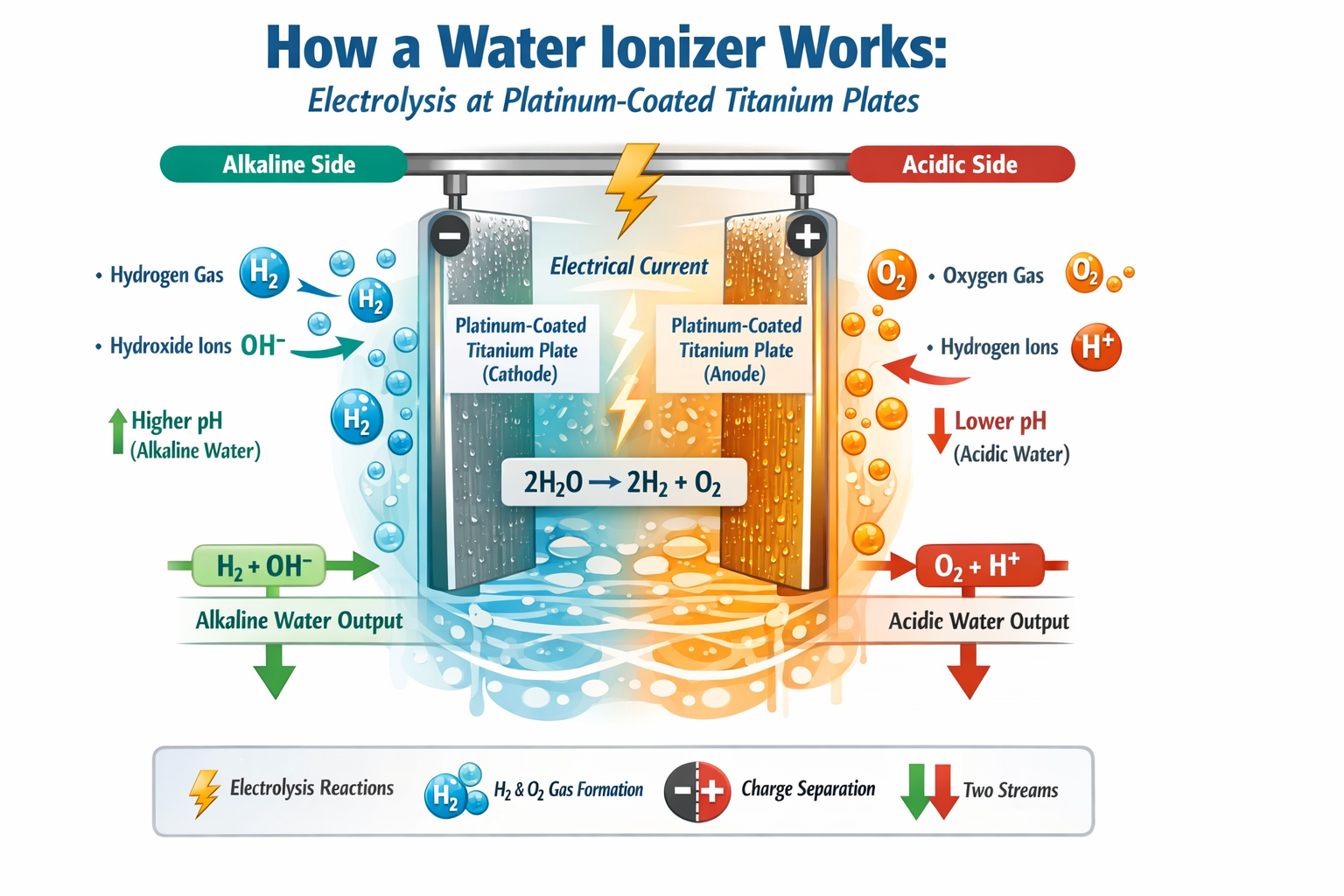
Section 6: Electrochemistry and Platinum Catalysis: How a Water Ionizer Works
Why the Materials Inside a Water Ionizer Matter
When people first learn about water ionizers, they often focus on the visible parts of the machine — the screen, the settings, the design, or the pH numbers listed in a brochure.
But the real work of a water ionizer happens out of sight.
Inside the machine is an electrolysis chamber containing metal plates. These plates are where the chemistry changes. They are the heart of the ionizer.
This is one reason the quality of the electrode system matters so much.
A water ionizer is not just a filter with a digital display. It is an electrochemical device. Its performance depends heavily on the quality of the materials used, the condition of the plates, and how efficiently electricity is transferred into the water.
What Electrochemistry Means in Plain English
Electrochemistry is the study of chemical changes caused by electricity.
In a water ionizer, electricity is applied to water as it moves across a series of plates. This creates chemical reactions that would not happen in ordinary filtered water alone.
In plain English, the machine uses electricity to encourage water and dissolved minerals to separate and react in specific ways.
That process leads to several measurable changes, including:
- generation of dissolved molecular hydrogen
- separation into alkaline and acidic streams
- changes in pH
- changes in oxidation-reduction conditions
- migration of minerals toward different sides of the chamber
These are not marketing ideas. They are the direct result of electrochemical reactions.
Why Platinum Is Used
Most quality water ionizers use titanium plates coated with platinum.
This raises an obvious question:
Why platinum?
Platinum is used because it is an excellent catalyst for electrolysis.
A catalyst is a material that helps a chemical reaction occur more efficiently without being consumed in the process.
In the case of a water ionizer, platinum helps the electrolysis reactions occur more effectively at the plate surfaces.
This matters because electrolysis is a surface event. The reactions happen where the water touches the plates. The better the surface properties of those plates, the more efficiently the machine can perform.
Why Titanium Is Used Under the Platinum
If platinum is so important, why not make the entire plate out of platinum?
The answer is practical.
Titanium provides a strong, durable, corrosion-resistant base metal. Platinum is then coated onto that titanium surface to provide the catalytic outer layer.
This combination gives the machine:
- strength
- corrosion resistance
- catalytic efficiency
- long-term durability
So when people talk about “platinum plates,” what they usually mean is platinum-coated titanium plates.
What Platinum Catalysis Actually Does
The platinum surface helps support the electrochemical reactions that produce changes in the water.
One of the key reactions on the alkaline side is:
2H2O + 2e- → H2 + 2OH-
This reaction helps explain two of the most important outcomes of ionization:
- H2 = dissolved molecular hydrogen
- OH- = hydroxide ions, which contribute to alkalinity
Platinum helps this reaction occur more efficiently by improving electron transfer at the plate surface.
In simpler terms, platinum helps the machine do its job better.
Why Plate Surface Matters So Much
Because electrolysis happens at the plate surface, the condition of that surface matters tremendously.
If the plate surface is clean and active, electrolysis can occur efficiently.
If the surface becomes coated with mineral scale, performance can drop.
This is especially important in areas with hard water, where calcium and magnesium can accumulate over time.
Even a very thin layer of mineral buildup can interfere with:
- direct contact between water and the catalytic surface
- efficient electron transfer
- hydrogen production
- stable electrolysis performance
This is one reason high-quality ionizers often include automatic cleaning systems.
Why Continuous Cleaning Matters
Many better ionizers are designed to reverse polarity or run cleaning cycles that help reduce scale buildup on the plates.
This matters because electrolysis depends on the quality of the active surface.
If calcium or other minerals form even a microscopic coating over the platinum, the water is no longer interacting with the catalytic surface as effectively.
That does not necessarily stop the machine from functioning, but it can reduce efficiency over time.
In practical terms, a well-maintained ionizer with effective cleaning cycles is more likely to:
- maintain hydrogen production
- maintain stable electrolysis
- perform more consistently over the long term
This is one of the hidden differences between higher-quality ionizers and cheaper machines that may look similar from the outside.
Electrode Quality Affects Real-World Performance
Two ionizers can both advertise multiple plates and alkaline water output, yet perform very differently in actual use.
That is because performance depends on more than plate count.
It also depends on:
- plate size
- plate spacing
- surface area
- electrical current control
- quality of the platinum coating
- water flow rate
- self-cleaning design
This helps explain why two machines with similar brochure claims may not produce the same hydrogen levels or the same long-term performance.
Electrochemistry is not just about having plates. It is about how well the whole system is designed and maintained.
Platinum Catalysis and the Two-Stream Process
The catalytic performance of the plates is also central to the machine’s ability to separate water into two streams.
As electricity flows through the water, the plates help create local conditions that encourage the separation of alkaline and acidic outputs.
This process involves:
- charge separation
- migration of ions
- gas formation
- changes in local pH conditions
The better the electrochemical environment inside the chamber, the more effectively the machine can carry out this separation.
So platinum catalysis is not just about hydrogen. It is about the entire electrochemical process that makes a water ionizer different from ordinary filtration.
What This Means for the User
From the user’s point of view, all of this comes down to a practical reality:
The quality of the plates and the quality of the electrolysis chamber directly affect the quality of the water the machine produces.
That influences:
- how consistently the machine produces alkaline water
- how effectively it produces dissolved hydrogen
- how well it maintains performance over time
- how much maintenance the machine may require
This is why serious evaluation of a water ionizer should go beyond cosmetic features and headline claims.
The true value of the machine lies in the unseen electrochemical system inside it.
Plain English Takeaway
A water ionizer works because electricity interacts with water at the surface of special metal plates.
Those plates are usually made of titanium coated with platinum.
Platinum helps the electrolysis reactions happen more efficiently, including the reactions that produce dissolved hydrogen and alkaline water.
If the plates stay clean and the system is well designed, the machine performs better. If mineral scale coats the plates, performance can gradually decline.
So when it comes to water ionizers, the materials inside the machine matter just as much as the features on the outside.
Conclusion
Electrochemistry is at the core of how a water ionizer works.
The electrode plates are not just passive parts. They are the active surfaces where the machine changes water through electricity.
Platinum catalysis helps make these reactions more efficient, while titanium provides the strength and durability needed for long-term use.
Understanding this helps explain why quality, design, and maintenance matter so much in the real-world performance of a water ionizer.
It also helps explain why the difference between one machine and another is not always visible from the outside. Often, the most important differences are happening inside the electrolysis chamber where the water meets the plates.
Section 7: Ionized Water vs. Hydrogen Water Machines

Two Technologies with a Shared Goal
As interest in hydrogen-rich water has grown, two main types of devices have emerged that produce water containing dissolved molecular hydrogen:
- water ionizers
- hydrogen water machines or generators
Although these technologies operate differently, they share a common goal: producing drinking water that contains dissolved molecular hydrogen.
What Water Ionizers Do
Water ionizers use a process called electrolysis.
Inside the ionizer, filtered water flows across a series of metal plates while a controlled electrical current is applied.
During this process several things happen simultaneously:
- water molecules split into hydrogen and oxygen
- dissolved molecular hydrogen forms in the alkaline stream
- minerals migrate toward different sides of the electrolysis chamber
- the water separates into alkaline and acidic outputs
What Hydrogen Water Machines Do
Hydrogen water machines focus primarily on dissolving molecular hydrogen into water.
Unlike ionizers, these systems generally do not separate water into two streams and usually keep the water close to its original pH.
The main goal of these machines is simply to add dissolved hydrogen gas to drinking water.
How the Two Systems Overlap
Both technologies can produce dissolved molecular hydrogen, which is the focus of most modern hydrogen water research.
Important Differences
- Ionizers separate water into alkaline and acidic streams
- Ionizers alter mineral distribution and pH
- Hydrogen machines mainly dissolve hydrogen gas
- Hydrogen machines usually leave mineral balance unchanged
Why Some People Use Both Technologies
Some people choose to use both a water ionizer and a dedicated hydrogen water machine because the two technologies emphasize slightly different aspects of water chemistry.
Water ionizers perform electrolysis, which separates water into two streams. During this process, minerals migrate within the electrolysis chamber and the water divides into an alkaline stream and an acidic stream. At the same time, dissolved molecular hydrogen is generated in the alkaline water.
Because electrolysis involves electrical fields, catalytic plate surfaces, and charge separation, some researchers and long-time users have suggested that the process may temporarily influence how water molecules organize at the microscopic level. While the full details of these effects are still being explored scientifically, the electrochemical environment created inside an ionizer is quite different from simply dissolving hydrogen gas into water.
For this reason, some people feel that freshly produced ionized water has characteristics that go beyond hydrogen content alone.
Hydrogen water machines, on the other hand, are designed primarily to dissolve hydrogen gas into water and often aim to produce higher concentrations of dissolved hydrogen.
As a result, some individuals choose to use:
- ionized water for daily hydration, cooking, and beverages
- hydrogen water machines when focusing specifically on hydrogen intake
In this way, the two technologies can be viewed as complementary rather than competing approaches.
Why Many People Use Ionized Water for Cooking and Hot Drinks
One practical question often arises when discussing hydrogen-rich water: what happens when the water is heated?
Molecular hydrogen is a very small gas molecule. When hydrogen-rich water is heated or vigorously stirred, much of the dissolved hydrogen gradually escapes into the air.
Because of this, heating hydrogen water for coffee, tea, or cooking will typically reduce the hydrogen concentration.
However, water produced by a water ionizer is not defined by hydrogen alone. During electrolysis, minerals migrate within the electrolysis chamber and the water separates into alkaline and acidic streams. These electrochemical changes influence the mineral balance and chemical environment of the water.
For this reason, many long-time ionizer users continue to use ionized water for:
- coffee and tea
- soups and broths
- cooking grains and vegetables
- everyday kitchen use
even though the hydrogen level may decline during heating.
Hydrogen water machines are typically used when the goal is to consume fresh hydrogen-rich water directly, while ionized water may still be used for a wider range of everyday applications.
The Importance of Fresh Water
Molecular hydrogen is a very small gas molecule and can gradually escape from water over time.
For this reason, hydrogen-rich water is generally best consumed soon after it is produced.
Plain English Takeaway
Water ionizers and hydrogen water machines use different technologies, but both aim to produce water containing dissolved molecular hydrogen.
Understanding these differences helps people evaluate which system best fits their hydration goals.
Section 8: Measuring Hydrogen in Water

Why Measuring Hydrogen Matters
As research into molecular hydrogen has expanded, an obvious question has emerged:
How do we know how much hydrogen is actually present in water?
Unlike minerals or pH, hydrogen gas is invisible and odorless. It dissolves into water without changing the appearance of the liquid.
Because of this, hydrogen-rich water can look exactly the same as ordinary water even when the dissolved hydrogen levels are very different.
For anyone studying hydrogen water seriously, measurement becomes important.
Why Hydrogen Is Difficult to Measure
Hydrogen is the smallest molecule in nature.
Because of its extremely small size, hydrogen gas moves easily through water and can gradually escape back into the air.
This means hydrogen levels can change depending on:
- how recently the water was produced
- how the water is stored
- temperature
- agitation or stirring
- exposure to air
Laboratory Methods for Measuring Hydrogen
In scientific research, several laboratory techniques are used to measure dissolved hydrogen.
- gas chromatography
- sealed headspace analysis
- specialized dissolved hydrogen sensors
These methods provide very precise measurements but require specialized equipment.
Practical Measurement Tools
Portable meters have been developed that allow users to measure dissolved hydrogen directly in water.
One example is the Trustlex ENH-2000 molecular hydrogen meter, which uses electrochemical sensing technology to detect hydrogen levels in water.
Devices like this allow users to verify hydrogen production and compare the performance of different hydrogen water systems.
Why Fresh Hydrogen Matters
Hydrogen measurements show that freshly produced water usually contains the highest hydrogen concentration.
Over time, especially when exposed to air, hydrogen can gradually escape from the water.
For this reason, hydrogen-rich water is generally best consumed soon after it is produced.
Plain English Takeaway
Hydrogen in water cannot be seen or tasted. Measurement tools make it possible to verify whether a system is producing hydrogen-rich water.
These measurements reinforce a simple principle: fresh hydrogen water consumed regularly is often more meaningful than extremely high concentrations taken occasionally.
Why Extreme Numbers Are Not the Goal
One final point is worth emphasizing.
In discussions about hydrogen water, it is easy to become overly focused on numbers. People sometimes assume that the highest possible hydrogen concentration must always be the most beneficial.
But current research suggests that molecular hydrogen may act largely through cellular signaling, helping activate protective pathways that regulate oxidative balance inside cells. Because signaling systems often operate at very small concentrations, extremely high hydrogen levels are not necessarily required.
For most users, the important question is simply whether a device produces measurable dissolved hydrogen in freshly prepared water.
Water ionizers and hydrogen water machines are typically tested during manufacturing to confirm that they generate hydrogen properly. Once operating correctly, routine use and normal maintenance are usually sufficient to maintain performance.
Some people enjoy measuring hydrogen levels themselves as part of exploring the science more deeply. For those interested in doing so, portable meters such as the Trustlex ENH-2000 molecular hydrogen meter can verify hydrogen levels directly in water.
However, measurement is not necessary for most users.
A simple way to confirm that a water ionizer is functioning properly is to test the alkaline and acidic streams with pH reagent drops. When the two streams show the expected pH separation, it confirms that the electrolysis process inside the machine is working as designed.
In the end, the goal is not to chase extreme numbers.
The goal is simple: clean water, produced consistently, containing measurable hydrogen and consumed regularly as part of normal hydration.
Further Reading & Video Options
I promised you further reading for those who want to explore the topics even further. Plus I produced several videos of this page for those who prefer multiple modalities for learning.
- Water Ionizer Use and Care
- Hydrogen Water & Ionized Water Research
- Water Ionizer Comparisons
- Several Part Video Series About This Page: The Science of Ionized Water, Hydrogen Water, and Water Structure
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 1
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 2
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 3
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 4
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 5
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 6
- The Science of Ionized Water, Hydrogen Water, and Water Structure Part 7
